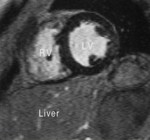
Case 1:
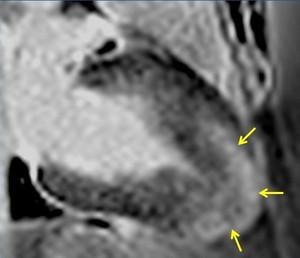
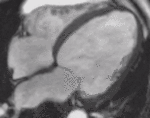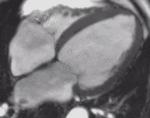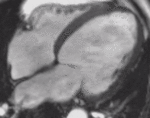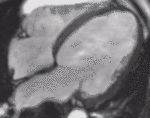
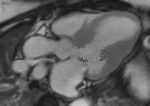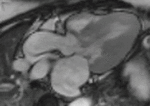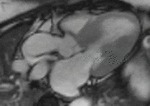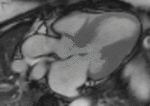
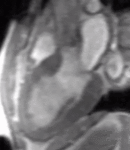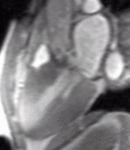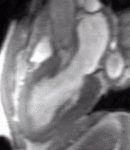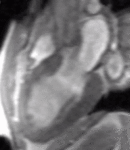
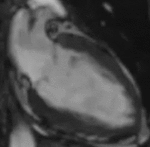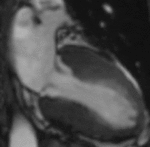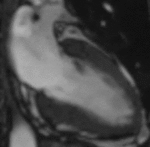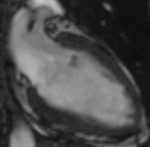
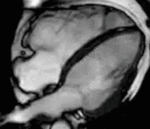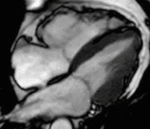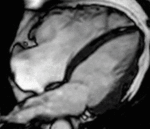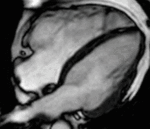
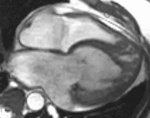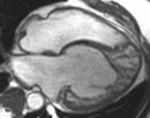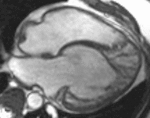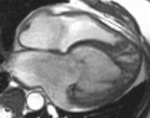
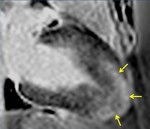
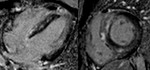
Fig. 1: Two-chamber viability image shows marked wall thickening and marked mesocardial and epicardial delayed enhancement at the LV apex, consistent with apical hypertrophic cardiomyopathy.
1.
Hypertrophic cardiomyopathy (HCM) (is the most common cardiomyopathy)
a.
Imaging features:
There are many different phenotypes of myocardial hypertrophy ranging from asymmetric to concentric to isolated hypertrophy of a papillary muscle.
Criteria for diagnosis include a wall thickness of ≥15 mm and a wall thickness ratio of ≥1.5 compared to the lateral wall.
The most common phenotype is asymmetric hypertrophy involving the interventricular septum.
Septal hypertrophy involving the left ventricle (LV) base may cause dynamic narrowing of the left ventricular outflow tract (LVOT),
in which case it is called hypertrophic obstructive cardiomyopathy (HOCM).
CMR can evaluate the extent of wall thickening in HCM,
especially.
CMR can demonstrate flow turbulence,
such as that seen during systole in the LVOT in patients with HOCM.
CMR can detect mitral regurgitation as well as systolic anterior motion of the mitral valve (SAM),
both of which are frequently seen in patients with HOCM.
(The mechanism of SAM is debated,
but the prevailing theory posits that flow acceleration due to dynamic LVOT obstruction pulls in the anterior mitral valve leaflet via the Venturi effect.) Viability imaging should be performed on all patients with HCM due to the possibility of fibrosis within the hypertrophied myocardium.
Up to 50% of patients with HCM demonstrate some fibrosis (as evidenced by late gadolinium enhancement),
which can be a nidus for fatal arrhythmias.
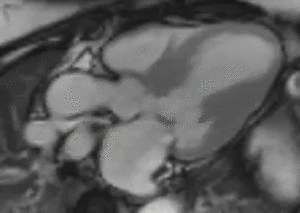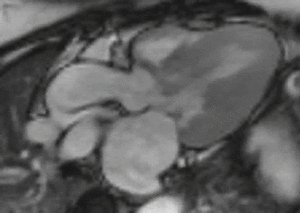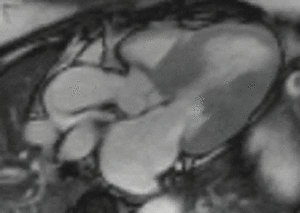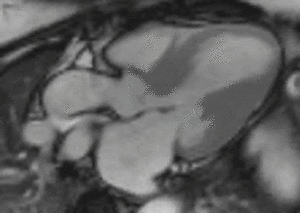
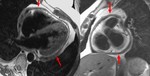
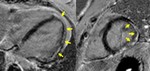
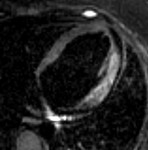
Fig. 2: LVOT bright blood cine b-SSFP loop in a patient with HOCM shows asymmetric septal hypertrophy, dynamic systolic narrowing and flow turbulence of the left ventricular outflow tract, SAM, and mitral regurgitation.
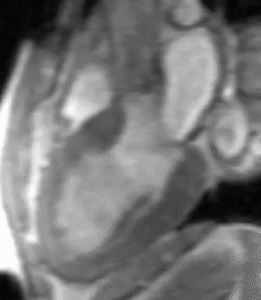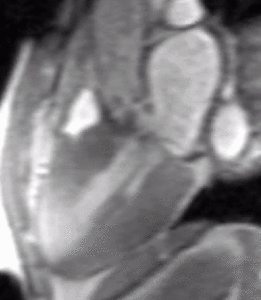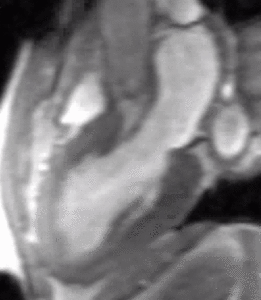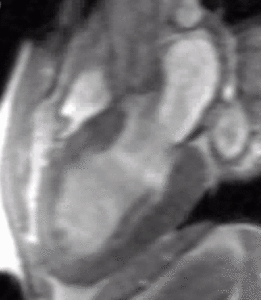
Fig. 3: LVOT bright blood cine gradient-echo loop in a patient with hypertrophic obstructive cardiomyopathy (HOCM) shows asymmetric septal hypertrophy, dynamic systolic narrowing and flow turbulence of the left ventricular outflow tract, systolic anterior motion of the mitral valve (SAM), and mitral regurgitation. Compared to b-SSFP, which has high SNR and excels at depicting cardiovascular anatomy (see figure 2), the partially refocused gradient-echo sequence used in this cine loop shows greater signal loss from systolic flow turbulence in the sub-aortic valve region and from the mitral regurgitant jet.
b.
Clinical Pearls:
HCM (if unrelated to hypertension or aortic stenosis) is an autosomal dominant condition that results in diastolic heart failure due to impairment of myocardial relaxation.
CMR can be used to follow patients after ventricular septal resection or percutaneous ablation.
A wall thickness ≥30 mm is an indication for ICD placement.
Case 2:
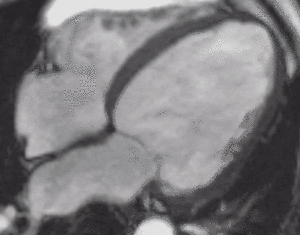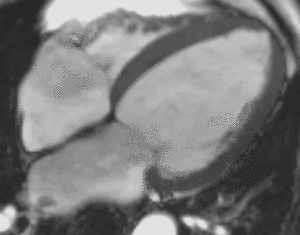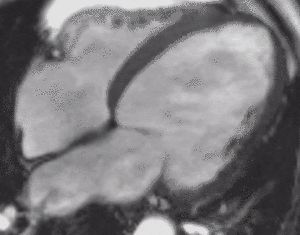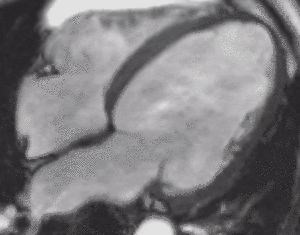
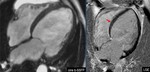
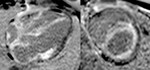
Fig. 4: Four-chamber bright blood cine loop showing markedly dilated left ventricle with severely decreased systolic function, but no regional wall motion abnormalities. Viability imaging showed faint diffuse mid myocardial enhacement (not shown). Findings are compatible with nonischemic dilated cardiomyopathy.
2.
Dilated cardiomyopathies (DCM)
a.
Imaging features:
Characterized by ventricular dilatation and global systolic dysfunction with impaired wall thickening and myocardial fiber shortening.
DCM can be associated with intracardiac thrombus.
The abnormalities seen in primary DCM can mimic those of ischemic cardiomyopathy.
CMR,
especially bright blood cine imaging and viability imaging,
can be used to differentiate the two entities.
DCM shows globally depressed systolic function,
but no regional wall abnormalities.
The latter are characteristic of old ischemic infarcts.
On viability imaging,
ischemic cardiomyopathy shows areas of subendocardial or transmural delayed enhancement in a coronary distribution (these typically co-localize with areas of abnormal wall motion).
In contrast,
DCM shows little or no delayed enhancement.
When present,
delayed enhancement in DCM is weak and diffuse and primarily involves the mid-myocardium.
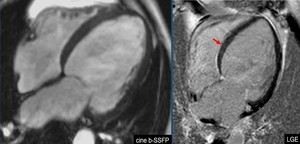
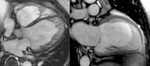
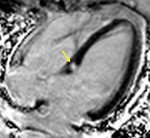
Fig. 5: A case of primary DCM showing dilated cardiac chambers. The left ventricle is most severely affected. The four-chamber bright blood cine loop show poor systolic function, but no regional wall motion abnormalities. Four-chamber PSIR viability imaging shows faint diffuse mid myocardial late gadolinium enhancement (red arrow).
b.
Clinical Pearls:
DCM is a common cause of congestive heart failure (CHF).
It is characterized by fibrosis and a reduced number of myocytes.
DCM is the most common primary cardiomyopathy.
About half of cases are idiopathic.
The remainder are due to a variety of etiologies,
including inflammation,
infection (viral or bacterial myocarditis),
infiltrative processes (sarcoid,
amyloid),
metabolic disorders (uremia,
hypocalcemia,
hypophosphataemia,
thyrotoxicosis),
toxins (alcohol,
cocaine),
chemotherapy (doxororubicin),
peripartum cardiomyopathy,
muscular dystrophies,
auto-immune cross reactivity (e.g.
late onset cardiomyopathy in Chagas disease),
and familial dilated cardiomyopathy.
Catheter angiography is recommended to exclude coronary artery disease and ischemic cardiomyopathy in a patient with newly diagnosed dilated cardiomyopathy.
Case 3:
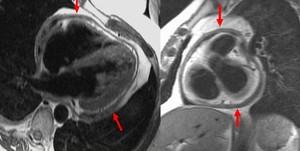
Fig. 6: Four-chamber (left panel) and short axis (right panel) T1-weighted dark blood images showing marked diffuse pericardial thickening (arrows) in a patient with constrictive pericarditis.
* Diagnosis is constrictive pericarditis
3.
Restrictive Cardiomyopathy (RCM)
a.
Imaging features:
RCM is characterized by small,
stiff,
thickened ventricles that impair diastolic filling.
This results in dilated atria and ultimately a dilated IVC.
The CMR features of RCM include normal LV size and systolic function,
diastolic dysfunction,
and biatrial enlargement.
b.
Clinical Pearls:
RCM causes diastolic heart failure.
Fibrosis or infiltration of the myocardium leads to increased wall rigidity and impaired diastolic relaxation.
RCM may be idiopathic or due to a variety of infiltrative processes such as amyloidosis,
hemochromatosis,
sarcoidosis (discussed below),
endomyocardial fibrosis,
scleroderma,
prior radiation therapy,
tumor invasion,
hypereosinophilic syndrome,
drugs (e.g.,
hydroxychloroquine [used to treat SLE,
rheumatoid arthritis and malaria],
methysergide,
anthracyclines),
Anderson-Fabry disease,
and glycogen storage diseases.
Clinically,
RCM is often difficult to differentiate from constrictive pericarditis.
The main role of CMR is to exclude constrictive pericarditis as the etiology of the diastolic dysfunction.
Constrictive pericarditis can be treated surgically,
whereas RCM has no effective treatment and a poor prognosis.
Endomyocardial fibrosis (and the related Loeffler’s endocarditis and hypereosinophilic syndrome) is a type of RCM in which there is extensive endocardial fibrosis involving the apex of one or both ventricles.
Apical thrombus or calcification may coexist.
The fibrosis appears as a dark endocardial rim at the apex on gradient echo sequences.
Case 4:
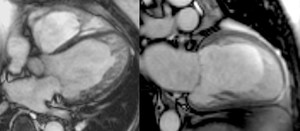
Fig. 7: Four-chamber (left panel) and two-chamber (right panel) bright blood cine b-SSFP frames in a patient with left ventricular non-compaction shows heavy trabeculation of the left ventricular wall. Note relative basal and septal sparing.
4.
Left ventricular non-compaction:
(LVNC,
also known as spongiform cardiomyopathy) is a developmental defect in embryologic formation of the left ventricle.
It is due to a failure of the left ventricle to form a solid (compacted) myocardium.
a.
Imaging features:
The left ventricle appears heavily trabeculated,
much like a right ventricle.
The compacted left ventricular wall is relatively thin.
Relative basal and septal sparing are common.
The CMR criteria for LVNC is a ratio of noncompacted to compacted myocardium exceeding 2.3:1 at end-diastole.
Viability imaging may show subendocardial delayed enhancement corresponding to myocardial fibrosis.
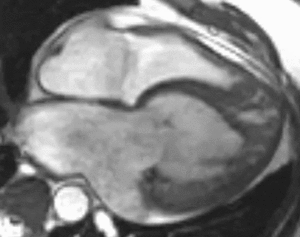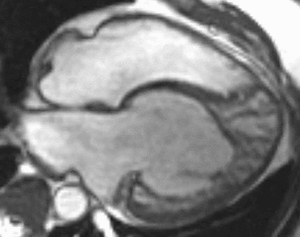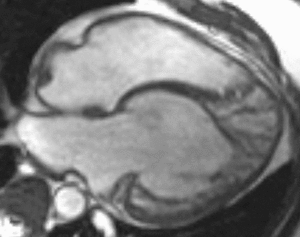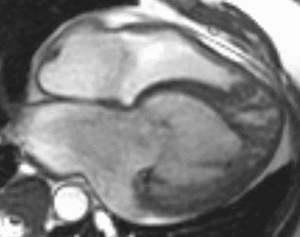
Fig. 8: Four-chamber bright blood cine loop demonstrates a dilated systemic ventricle with heavy trabeculations and decreased systolic function. The ipsilateral atrioventricular valve is regurgitant. The trabeculations suggest left ventricular non-compaction, but the real diagnosis is L-transposition of great arteries (L-TGA, also called congenitally corrected transposition) with a failing systemic right ventricle. A moderator band in the systemic ventricle provides a hint. The relative positions of the aortic and pulmonic valves (the former is anterior and to the left) clinch the diagnosis.
b.
Clinical Pearls:
Patients have an increased risk of adverse cardiac events,
including arrhythmias,
heart failure,
thrombus formation,
stroke and sudden cardiac death.
The only definitive treatment of LVNC is cardiac transplantation.
Case 5:
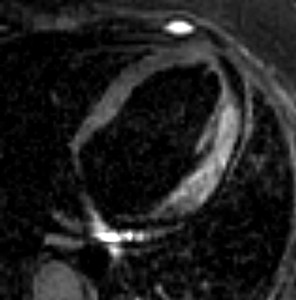
Fig. 9: Axial T2-weighted dark blood image shows hyperintensity in the lateral LV wall. consistent with myocardial edema. The patient had viral myocarditis, but an acute myocardial infarction involving the lateral wall could have a similar imaging appearance.
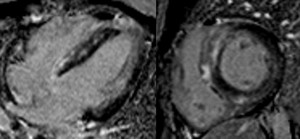
Fig. 10: Four-chamber (left panel) and short axis (right panel) viability images show patchy epicardial and mid myocardial delayed enhancement with septal predominance. The distribution of enhancement does not follow coronary artery territories. The patient had giant cell myocarditis, a rare and serious disease. Patient died shortly after the CMR.
5.
Myocarditis
a.
Imaging features:
Inflamed myocardium appears hyperintense on T2-weighted images due to edema and shows increased early enhancement after gadolinium administration on T1-weighted images due to increased blood flow.
Viability imaging demonstrates mid-myocardial and epicardial enhancement,
often in a patchy pattern that does not follow coronary artery territories.
Regional or global wall motion abnormalities are common,
but nonspecific.
b.
Clinical Pearls:
The most common cause of myocarditis is viral infection,
usually with Coxsackievirus B.
Other infections include echovirus,
arbovirus,
bacterial (e.g.
Corynebacterium diphtheriae,
Streptococcus pyogenes,
Staphylococcus aureus,
Borrelia burgdorferi),
fungal (e.g.
Candida),
or parasites (e.g.
Trypanosoma cruzi).
Non-infectious etiologies include drugs (e.g.,
antibiotics,
anti-tuberculous agents,
anticonvulsants,
anti-inflammatories,
diuretics,
recreational drugs),
radiation,
transplant rejection,
giant cell myocarditis (rare),
and systemic lupus erythematosus.
Case 6:
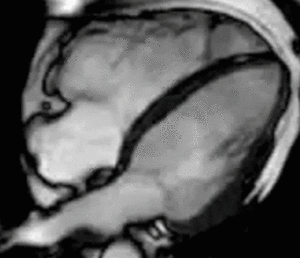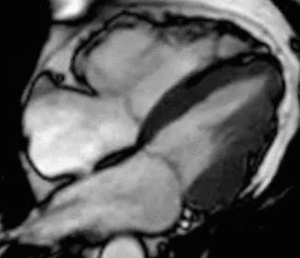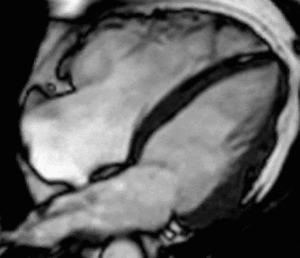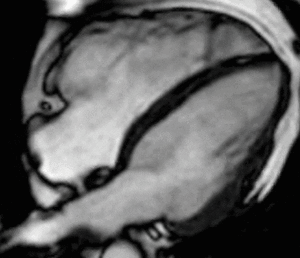
Fig. 11: Four-chamber bright blood cine loop showing a dilated right ventricle (RV) with modestly decreased systolic function and a localized aneurysm of the basal free wall. Findings are consistent with ARVC, but imaging alone is insufficient to establish the diagnosis.
6.
Arrhythmogenic cardiomyopathy
(formerly called arrhythmogenic right ventricular dysplasia or cardiomyopathy,
as it was thought to affect only the right ventricle) is a familial cardiomyopathy in which there is fibrous or fibro-fatty replacement of ventricular myocytes.
The ventricles show focal contraction abnormalities and/or aneurysm formation.
Although right ventricular involvement predominates,
the eft ventricle can be involved in up to three quarters of patients.
a.
Imaging features:
Diagnosis is usually difficult and depends on major and minor criteria gathered from multiple sources,
including family history,
genetic testing,
EKG,
imaging,
and biopsy.
No single modality or source is sufficient to establish the diagnosis.
Imaging findings,
including CMR,
may contribute at most one major or one minor criterion.
Major imaging criteria for diagnosis include severe global or regional dysfunction of the RV (with little or no LV impairment),
global RV dilatation,
or localized RV or right ventricular outflow tract (RVOT) aneurysm.
The presence of myocardial fat is no longer a criterion as fat can be seen in normal individuals with aging.
Delayed enhancement of the right ventricle suggests infiltration with fibrotic tissue,
a finding that would be consistent with arrhythmogenic cardiomyopathy.
b.
Clinical Pearls:
Fibro-fatty infiltration interferes with electrical conductance in the myocardium,
predisposing to ventricular arrhythmias and sudden death.
Patients therefore require ICD placement once the diagnosis is confirmed.
Right ventricular dilatation and right sided heart failure can occur as well.
Case 7:
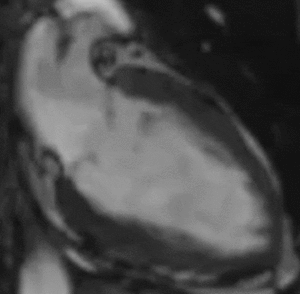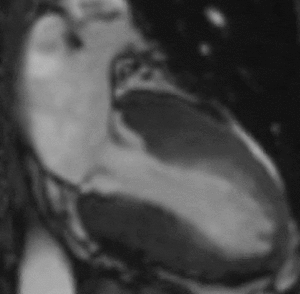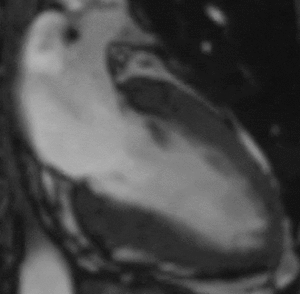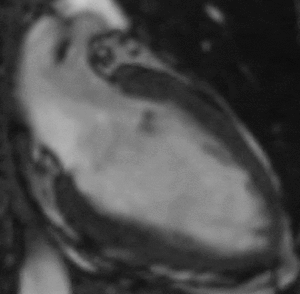
Fig. 12: Two-chamber bright blood cine loop demonstrating an akinetic left ventricular apex. Viability imaging was normal (not shown). Findings are typical of Takotsubo cardiomyopathy.
7.
Takotsubo cardiomyopathy
(also called Catecholamine induced cardiomyopathy,
or broken heart syndrome).
a.
Imaging features:
There is characteristic ballooning of the cardiac apex.
The shape of the heart resembles a Japanese octopus pot,
hence the name takotsubo.
Four distinct patterns of dyskinesia and ballooning are described: apical (most common),
biventricular,
mid-ventricular and basal.
Myocardial edema,
as evidenced by T2 hyperintensity,
may co-localize in areas of abnormal wall motion seen on cine MRI.
First-pass perfusion and viability imaging: Both are usually normal,
which differentiates takotsubo cardiomyopathy from anterior STEMI.
b.
Clinical correlates:
Left ventricular dysfunction is usually self-limited.
More common in older women in the setting of acute emotional stress.
Clinical presentation can mimic acute myocardial infarction.
Patients may present with chest pain,
abnormal ECG,
and elevated cardiac enzymes.
Cardiac catheterization is normal.
Case 8:
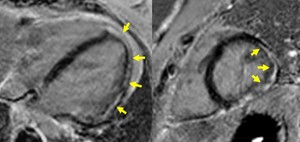
Fig. 13: Four-chamber (left panel) and short axis (right panel) PSIR viability images in a patient with Duchenne Muscular-Dystrophy show epicardial and mesocardial delayed enhancement in the lateral and inferior LV walls. The enhancement pattern is nonischemic.
8.
Duchenne Muscular-Dystrophy (DMD):
a.
Imaging features:
The cardiac involvement is typically in inferolateral wall and the basal inferior and anterolateral region of the left ventricle.
Late gadolinium enhancement (LGE) shows myocardial fibrosis,
which is typically transmural.
LGE occurs early,
is progressive and increases with both age and decreasing LV ejection fraction.
b.
Clinical Pearls:
Majority of DMD patients have cardiomyopathy by third decade,
but overt heart failure may be absent.
Cardiomyopathy is the main cause of death in DMD.
Case 9:
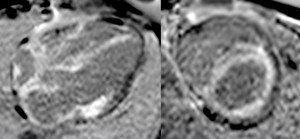
Fig. 14: Four-chamber (left panel) and short axis (right panel) PSIR viability images in a patient with cardiac amyloidosis show extensive transmural delayed enhancement. Note involvement of the RV free wall and both atria. The enhancement pattern could be confused for ischemic scar, but the distribution is clearly nonischemic because it does not follow coronary artery territories.
9.
Amyloidosis:
a.
Imaging features:
Ventricular wall thickening with diffuse transmural or subendocardial delayed enhancement.
There may also be thickening of the atrial septum or right atrial posterior wall.
b.
Clinical Pearls:
Amyloidosis involves the myocardium in nearly all cases of primary amyloidosis,
but is less common in secondary amyloidosis.
Cardiac involvement often presents as a restrictive cardiomyopathy.
Concentric left ventricular wall thickening in amyloidosis can be confused with concentric HCM.
Fortunately,
ECG will help to resolve the confusion: R-wave voltages are typically increased in HCM and decreased in cardiac amyloidosis.
Case 10:
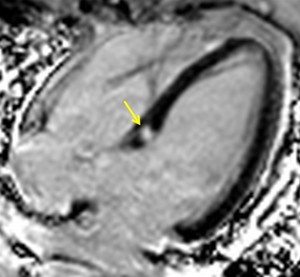
Fig. 15: Four-chamber PSIR viability image in a patient with sarcoidosis shows a nodular delayed enhancing granuloma in the basal septum.
10.
Sarcoidosis:
a.
Imaging features:
Focal or nodular mid-myocardial hyperintensity on T2 weighted images.
Focal myocardial thickening may also be present.
On viability imaging,
sarcoid granulomas typically demonstrate a patchy mid myocardial,
epicardial or transmural distribution.
b.
Clinical Pearls:
Sarcoidosis involves the heart in about 25% of patients,
but only 5-10% of patients are symptomatic.
Nonetheless,
cardiac involvement is probably an important contributor to sarcoidosis-related mortality.
The clinical spectrum of cardiac sarcoidosis includes arrhythmias including sudden death (most common) and heart block,
congestive heart failure,
angina pectoris,
palpitations and syncope,
and ventricular aneurysm formation.
Case 11:
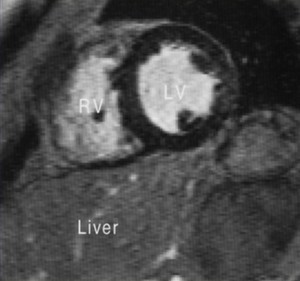
Fig. 16: Mid short axis T2*-weighted GRE image in a patient with genetic hemochromatosis shows diffuse signal loss in the myocardium due to abnormal increased iron deposition.
11.
Hemochromatosis:
a.
Imaging features:
Role of MRI is to identify and quantify iron deposition.
Iron deposition in the heart is predominantly subepicardial.
Due to the paramagnetic properties of iron deposits,
signal loss is seen on both T1 and T2 weighted images.
Iron deposition in the liver causes hypointensity on in-phase T1,
T2 and T2*-weighted images on MRI.
b.
Clinical Pearls:
Hemochromatosis can be either primary (genetic) or secondary to severe anemia,
chronic liver disease,
or multiple blood transfusions.
The disease is characterized by iron deposition in many tissues throughout the body,
including the heart,
liver,
pancreas,
skin,
and joints.
Clinical manifestations of cardiac hemochromatosis depend on the extent of iron deposition and include dyspnea,
edema,
palpitations,
and other features of congestive heart failure.
12.
Anderson–Fabry disease:
An X-linked lysosomal storage disorder caused by a deficiency of the enzyme α-galactosidase A (α-Gal A),
results in abnormal increased cellular storage of the enzyme's substrate globotriaosylceramide (Gb3) and related glycosphingolipids.
a.
Imaging features:
Focal myocardial fibrosis and concentric left ventricular hypertrophy.
Thickening of the cardiac valves is seen in 25% of patients.
b. Clinical Pearls:
Fabry disease is a multisystem disorder.
50% of patients have cardiac involvement (called Fabry cardiomyopathy),
an important cause of disease-related mortality.
Treatment include enzyme substitution (hydroxylase alpha-galactosidase) which is efficacious in correcting the metabolic deficit.
In men,
the condition is fatal within 50 years without treatment.
Interestingly,
heterozygous women may be symptomatic and thus are not just carriers.